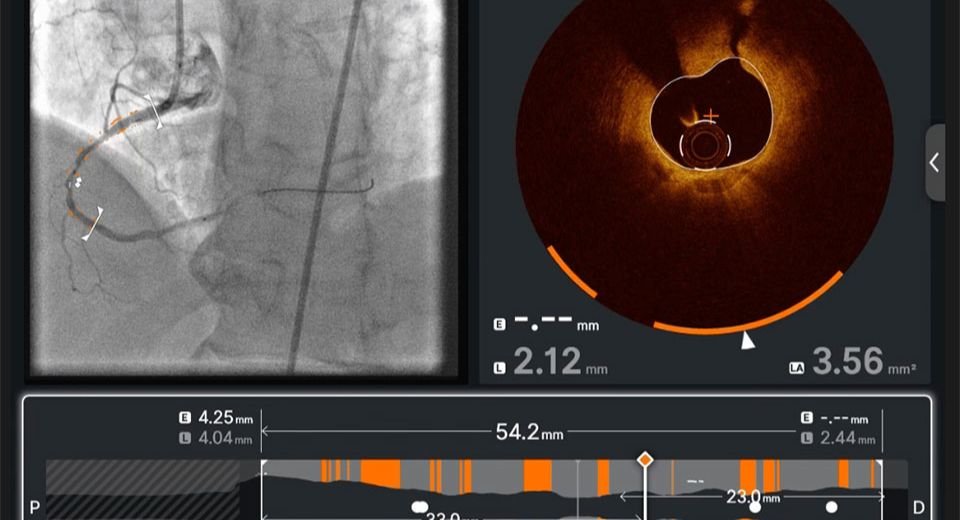
May 3, 2026: The US Food and Drug Administration has cleared Abbott Laboratories’ new artificial-intelligence-powered software for coronary imaging, enabling doctors to have real-time planning guidance while they perform minimally invasive procedures to open blocked heart arteries.
The Ulteron 3.0, an advanced optical coherence tomography (OCT) system, also got the CE mark, indicating that the product complies with essential EU health, safety, and environmental protection legislation, allowing it to be sold throughout the European Economic Area, according to a company statement.
Abbott’s technology allows physicians to use OCT images to view the detailed structure of the blockage, and its artificial intelligence assesses the type of plaque causing it, helping the doctor choose the best location and stent size to improve blood flow. The platform offers a fully integrated workflow to support faster, more precise procedures.
“Ultreon 3.0 represents a pivotal step forward in how we treat and ultimately care for our patients,” said Evan Shlofmitz, DO, interventional cardiologist, director of interventional cardiology, St. Francis Hospital and Health Center in Roslyn, New York.
“This next-generation platform, combining imaging and AI, doesn’t just improve upon existing technology — it leapfrogs it.”
Kidney disease
The software provides higher-resolution imaging than intravascular ultrasound while requiring little or no contrast. Reducing contrast use may benefit certain patients with kidney disease, who account for roughly 25% of individuals with coronary artery disease.
It helps doctors acquire images of inside blood vessels and enables them to efficiently plan procedures, specifically tailored for patients with complex coronary disease, according to the statement.
“Every second and every decision matters when treating patients who have blockages in their coronary arteries, and Ultreon 3.0 enables faster, more informed clinical choices that can have a real impact on patients,” said David M. Leistner, interventional cardiologist, Clinic for Cardiology and Angiology, Charité Campus Benjamin-Franklin (CBF) in Berlin, Germany.
Complex percutaneous coronary intervention is a specialised procedure used to treat severe coronary artery disease. It involves treating challenging lesions—or plaques—that have built up in a patient’s coronary arteries. Some patients will require multiple stents to address the lesions. More than 600,000 people in the US and more than 885,000 in Europe undergo the procedure each year.