HQ Team
March 25, 2026: Pfizer Inc. and France-based Valneva SE‘s investigational vaccine has shown 70% efficacy in preventing Lyme disease in individuals aged five and above.
The Saint-Herblain-headquartered Valneva and Pfizer stated that although the end-stage study did not meet the primary endpoint of reliability, they planned to make submissions to regulators.
“The pre-determined statistical criterion (95% confidence interval lower bound >20) was not met in the first pre-specified analysis (primary endpoint),” according to a joint statement.
For the main result (the primary endpoint) to be officially considered a “success,” the lowest possible value for the vaccine’s effectiveness had to be above 20%. If it dips to 20% or lower, the study technically misses the statistical target — even if the vaccine performed well overall.
‘Meaningful level of protection’
The researchers didn’t look at the average effectiveness of the results (which was actually much higher — around 70%+). They also considered the worst-case scenario that was still statistically believable. That “worst-case but still believable” number is called the lower bound of the 95% confidence interval.
The companies stated that during the study, fewer people got Lyme disease than the researchers had expected. Because of this, the main analysis did not quite meet the statistical goal they had set in advance.
However, the vaccine still showed a clear and meaningful level of protection. In a second planned analysis, the results met the statistical target, and the lower bound went above 20%, according to the statement.
Lyme disease (also called borreliosis) is an infection you can get from the bite of a deer tick. A bacterium called Borrelia burgdorferi causes the infection. Lyme disease is the most common tick-borne infectious disease in the US, according to the Cleveland Clinic.
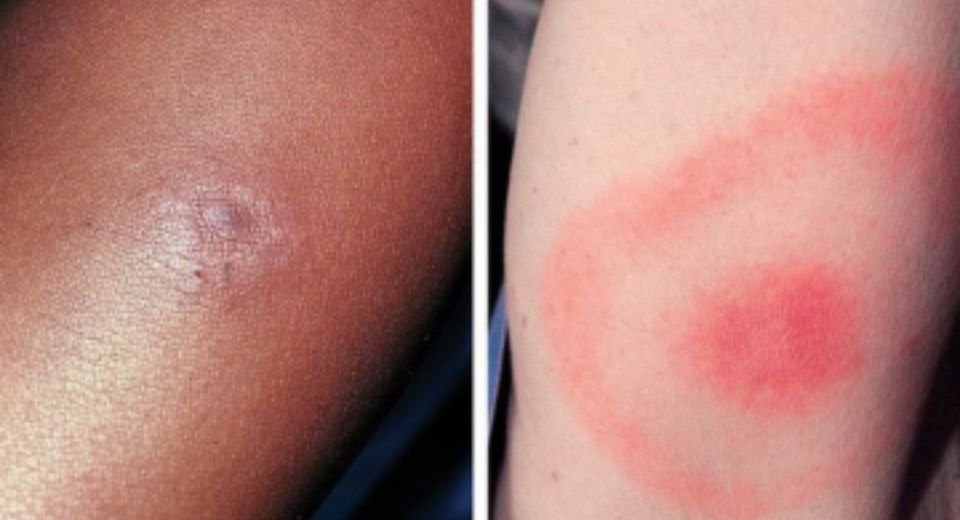
Bullseye rash
Initial symptoms of Lyme disease often include a “bullseye” rash, fever and fatigue. Without treatment, the infection can spread to many parts of the body. It can lead to complications affecting the nervous system, heart, and joints.
The bullseye rash starts as a small, red spot at the site of the tick bite. This can happen at any time between one and four weeks after the bite. The spot expands within days or weeks, forming a circular, triangular or oval-shaped rash.
The rash may look like a bullseye, with a red ring surrounding a clear centre. It can range in size from a dime to the width of your body. Not everyone with Lyme disease develops this rash. It appears in about seven to eight out of 10 infected people.
“Lyme disease can cause potentially serious consequences – where individuals and families face symptoms that can disrupt daily life, work, and long-term health – and there is currently no vaccine available,” said Annaliesa Anderson, PhD, Senior Vice President and Chief Vaccines Officer, Pfizer.
‘Debilitating’
“The efficacy shown in the study of more than 70% is highly encouraging and creates confidence in the vaccine’s potential to protect against this disease that can be debilitating.”
Thomas Lingelbach, CEO and Board member of Valneva, said: “These results bring us a step closer to our goal of delivering a much-needed vaccine to help protect against Lyme disease.”
According to the Centers for Disease Control and Prevention, about 476,000 people in the US are diagnosed and treated each year with Lyme disease, and 132,000 cases are reported annually in Europe from countries with surveillance systems.