HQ Team
March 7, 2024: The FDA has approved Pfizer’s Besponsa drug for pediatric patients aged one year and above to treat acute leukaemia.
Pfizer’s application was granted a priority review and orphan drug designation by the US drug regulator.
The efficacy of the medicine was evaluated in 53 pediatric patients one year and older with relapsed or refractory CD22-positive B-cell precursor acute lymphoblastic leukaemia.
The study’s primary endpoints were the complete remission rate, duration of complete remission, and minimal residue disease negativity.
In all patients, 42% achieved complete remission and the median duration of the remission was 8.2 months or 95%, according to an FDA statement.
Abdominal pain, headache
The minimal residue disease negativity rate in patients with complete remission was 95.5% and it was 86.4% based on real-time quantitative reverse transcriptase polymerase chain reaction — a test used for quantification of specific mRNAs.
The regulatory agency recommended a starting dose of 1.8 mg/m2 per cycle, given as 0.8 mg/m2 on day 1, 0.5 mg/m2 on day 8, and 0.5 mg/m2 on day 15 of cycle 1.
The most common adverse reactions including laboratory abnormalities, were thrombocytopenia, pyrexia, anemia, vomiting, infection, hemorrhage, neutropenia, nausea, leukopenia, febrile neutropenia, increased transaminases, abdominal pain, and headache.
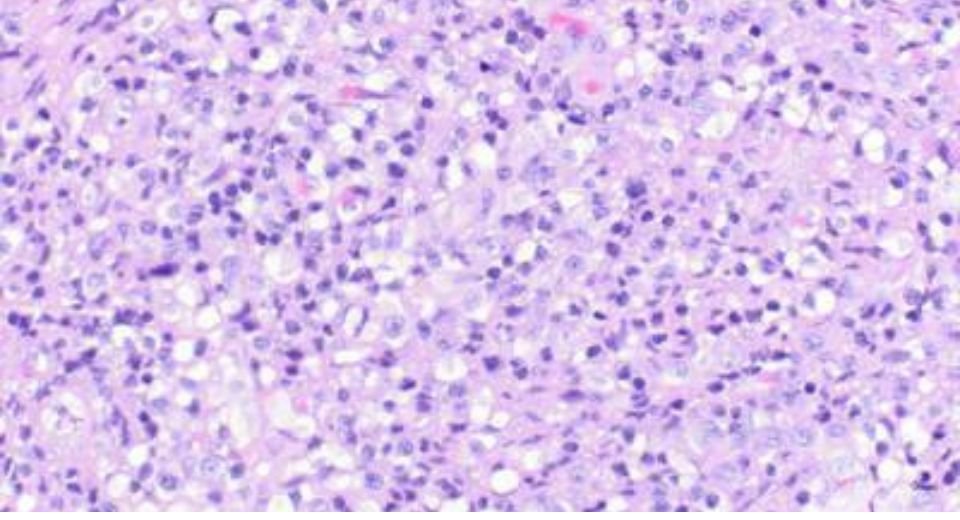
Acute lymphoblastic leukaemia is a type of cancer of the blood and bone marrow that affects white blood cells. It is the most common childhood cancer. It occurs when a bone marrow cell develops errors in its DNA.
Symptoms may include enlarged lymph nodes, bruising, fever, bone pain, bleeding from the gums and frequent infections. Treatments may include chemotherapy or targeted drugs that specifically kill cancer cells.
Survival rates are lower in adults, but they are improving. The five-year relative survival rate for acute lymphoblastic leukaemia is 69%. The statistics further break down to 90% in children and 30-40% in adults, according to the University Kansas Cancer Center.